|
12, 13 It has been demonstrated that in several cell lines, including HeLa cells used for the FRAP experiments described above, mitochondria exist as a heterogeneous mixture of elongated, tubular and short, vesicular forms, which may differ within one cell in the Δ Ψ m, the rate of permeability transition pore (PTP) activation and the dynamics of Ca 2+ sequestration. In addition to their appearance as network-like, functionally united organelles, mitochondria also can be morphologically and functionally independent within cells. In yeast, serial sectioning and reconstruction of electron micrographs as well as experiments with organisms expressing mito-GFP revealed also the presence of a single branching mitochondrion. Genetic complementation, likely achievable due to the dynamic network-like behavior, may represent a mechanism of repairing mtDNA mutations, and consequently maintain properly functioning oxidative complexes within the cell. mtDNA is prone to mutations that accumulate in aging individuals. 8 Introduction of mitochondria bearing mtDNA mutations that inactivate cytochrome c oxidase (COX) into cells with wild-type mitochondria, resulted in dynamic mixing of matrix and membrane proteins, as well as mtDNA transfer, between normal- and COX-activity-deficient organelles so that all mitochondria within the cell displayed comparable, high COX activity. 6, 7 Moreover, when fluorescence recovery after photobleaching (FRAP) has been applied, photobleaching of mito-GFP within a small area of the cell resulted in the sequential redistribution of the mito-GFP from unaffected portions of the cell, 5 suggesting lumenal continuity of the mitochondria within network(s) over broad areas of the cell.įrequent fusion and fission within a dynamic network may be an efficient means of intermitochondrial DNA (mtDNA) complementation through exchange of genomes between fusing mitochondria. 6, 7 High connectivity of these organelles within the cell has been supported by the fact that, in cells stained with mitochondria specific, membrane potential (Δ Ψ m)-sensitive fluorescent probes, depolarizing stimuli targeted to a small area of the mitochondrion, leads to the collapse of the Δ Ψ m within the network over the length of tens of micrometers. Several other lines of evidence suggest that, at least under some conditions, mitochondria in mammalian cells can form physically interconnected networks 6, 7 that may represent an efficient system to deliver energy, or channel calcium between different areas of the cell. 4 In addition, a network of interconnected, tubular mitochondria has been detected by high-resolution 3D imaging of mitochondria expressing green fluorescent protein targeted to the mitochondrial matrix (mito-GFP). The presence of dynamic, tubular, 28 nm junctions connecting cristae to the IMM was found by serial sectioning and EM tomography. 1, 2, 3 The classical ‘textbook’ view, based mainly on two-dimensional electron micrographs, of mitochondria as bean-shaped structures with a highly folded inner membrane (IMM) forming mitochondrial cristae enveloped by a relatively uncomplicated outer membrane (OMM) has been challenged since the introduction of 3D reconstitution technologies. 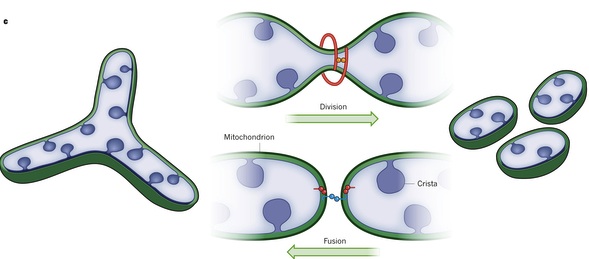
Imaging of living cells stained with vital, cationic fluorescent probes, such as Rhodamine 123 and JC-1, or green fluorescent proteins specifically targeted to mitochondria revealed that mitochondria are highly dynamic, motile and undergo frequent shape changes ( Figure 1). Mitochondria are dynamic organelles that can change in number and morphology within a cell during development, the cell cycle or when challenged with various toxic conditions. Dynamics of mitochondria in living cells: fission and fusion maintain homeostasis
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
